A CMMS to streamline workflows, reduce costs, and boost productivity
IMPROVE YOUR PRODUCTIVITY
Are you looking for a reliable solution to improve operational asset management and reduce maintenance costs? Corim® provides a powerful tool for industrial maintenance, helping to streamline asset management and enhance operational efficiency. By centralizing maintenance processes, Corim® enables better planning, tracking, and execution of maintenance tasks, reducing downtime and boosting productivity.
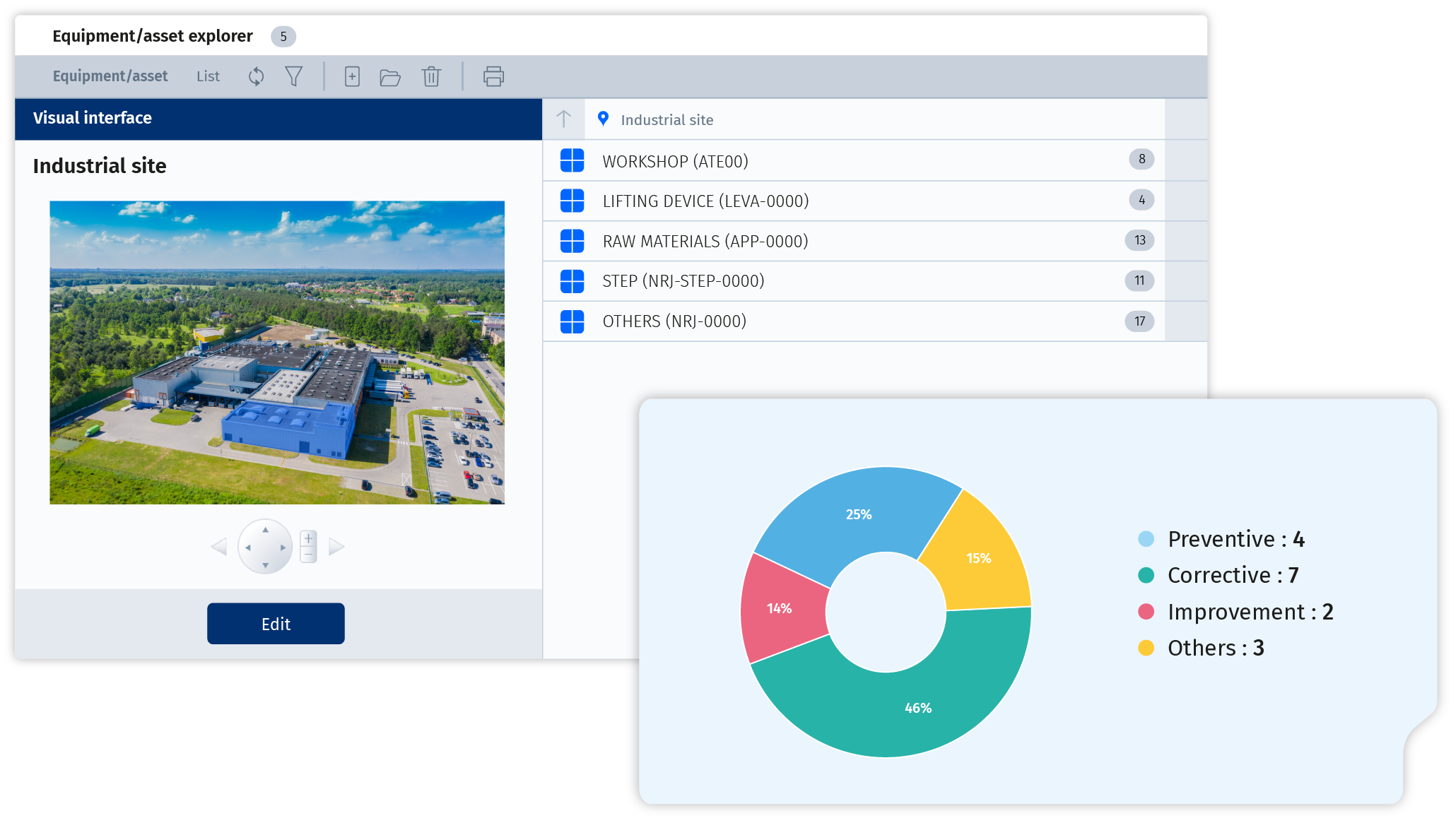
Wherever you are, access the full knowledge base of your equipment and assets: technical documents, manuals, schematics, history, criticality, and prioritized interventions. With a structured and precise database, you can act efficiently and make informed decisions.
Corim® centralizes all maintenance activities, offering tools to assign, track, and document tasks with precision. Technicians can access detailed instructions, historical records, and real-time updates, ensuring that every maintenance activity is executed efficiently and aligns with the organization’s operational goals.
increase in productivity
more work orders completed
improvement in input quality
PREVENTIVE MAINTENANCE SOFTWARE
Corim® helps you manage preventive maintenance for optimum equipment performance while reducing unplanned downtime. The system enables you to plan and execute tasks efficiently, based on predefined intervals or usage thresholds. Features such as the maintenance planning program and industrial preventive checklists help streamline operations, ensuring that essential inspections and maintenance are carried out on schedule.
PLANNED & REACTIVE MAINTENANCE
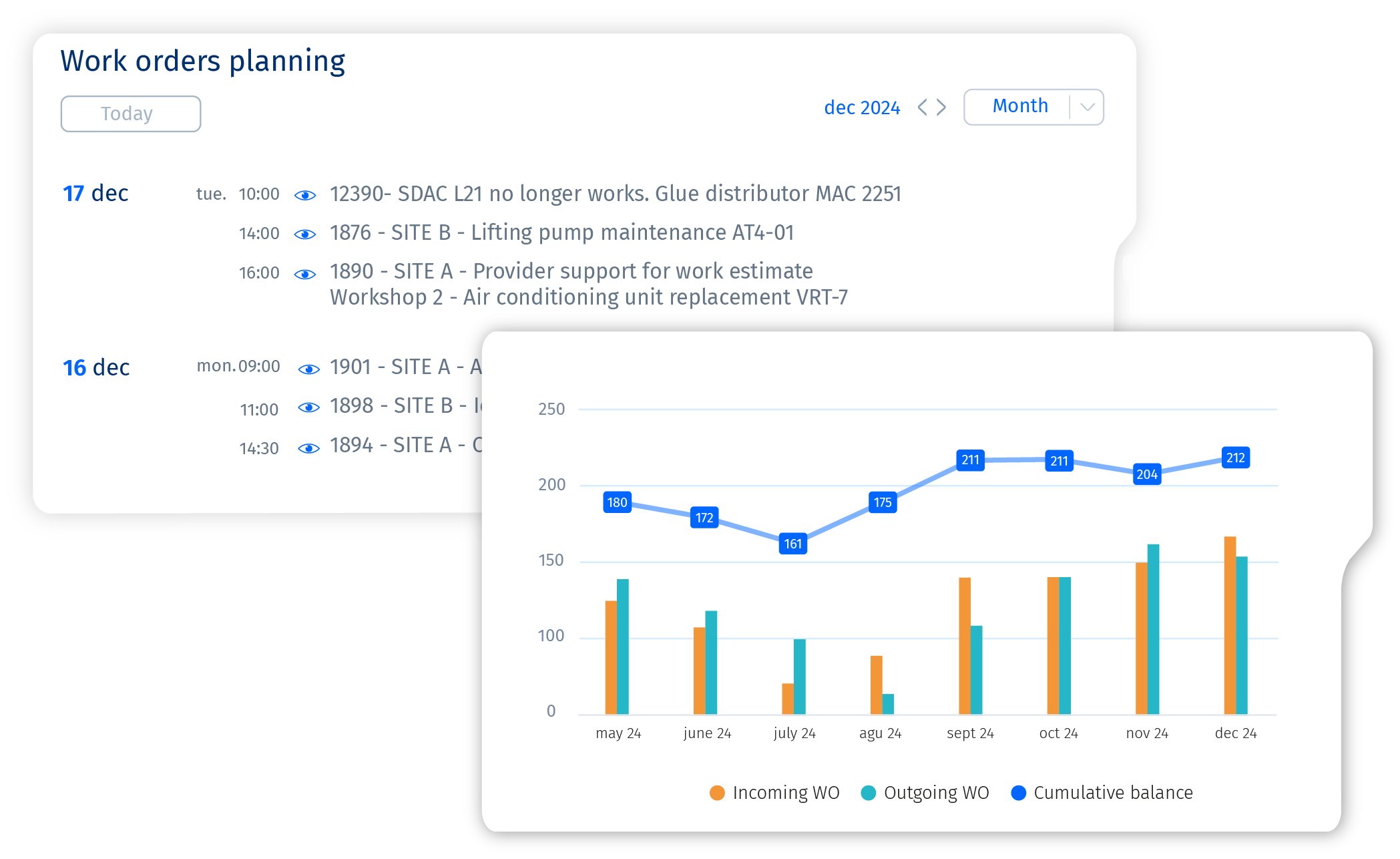
Planned maintenance is essential for preventing unexpected downtime and ensuring the long-term reliability of your equipment. Corim® is a powerful CMMS software that simplifies scheduling and managing maintenance activities, enabling managers to plan interventions efficiently. This ensures that critical tasks are completed on time—with the right materials and personnel—before they lead to costly breakdowns.
With automated scheduling tools, maintenance tasks can be planned at regular intervals or based on equipment usage. Features such as Maintenance Scheduling Programs and Industrial Preventive Checklists help ensure that inspections and servicing are performed periodically, reducing the risk of unexpected failures and prolonging asset lifespan.
When unplanned issues arise, Corim® facilitates a structured approach to reactive maintenance by prioritizing tasks, allocating resources effectively, and ensuring rapid response times. This minimizes disruptions and restores equipment functionality as quickly as possible.
"Today, we don't know how we could live without it. The reporting and information we obtain from it are essential."
Jean-Philippe LLORET, Maintenance manager and CMMS administrator - PETZL
.webp)
MAINTENANCE APPLICATION
Corim Touch is the essential maintenance app for technicians. By connecting your Corim® CMMS to Corim Touch, you can communicate in real time with your field teams, providing information on planned interventions and assistance with tasks. You benefit from data collected by your maintenance teams, with no risk of error or omission. Intervention reports, which are precise and easy to fill out, can be completed by voice, enriching your maintenance database.
With the maintenance app, you guarantee traceability of interventions, operationnal asset management, better team organization, time savings between interventions and a complete equipment history.
Equipment technical files are accessible anywhere, anytime, both online and offline. Your assets are easily identified via QR Code, and inventory status can be consulted remotely. For improved efficiency and responsiveness, you can maintain a constant connection with your teams and keep them promptly informed about any necessary work through push or SMS notifications.

SKILLS TRANSFER
One objective: to master your Corim® applications quickly and sustainably, when starting up a project or for a specific theme. Corim solutions offers skills transfer by profile, by functional area, and customized coaching, either face-to-face or remotely. Tailor-made, they are approached differently depending on the audience and the expected content. The sessions bring together identical profiles, and only the functionalities decided beforehand, profile by profile, are shown. The database is administered and personalised beforehand, so that participants are completely immersed in their Corim® environment.
Qualiopi certification
Training for French companies: as Corim solutions has been Qualiopi-certifed for its trainings since 2022, OPCO agencies can bear some training costs, for French customers only.